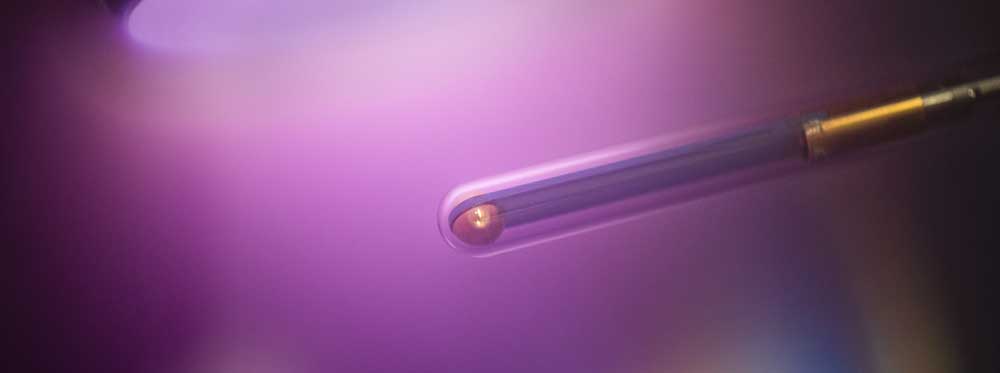
A3 - Plasma SourcesPlasma Bullet JetA plasma bullet jet is developed to monitor post-plasma |
- Details
Professor Uwe Czarnetzki Receives ISPlasma Special Recognition Award
 We are pleased to announce that Professor Uwe Czarnetzki has received the ISPlasma Special Recognition Award in honor of his outstanding and sustained contributions to the ISPlasma conference series.
We are pleased to announce that Professor Uwe Czarnetzki has received the ISPlasma Special Recognition Award in honor of his outstanding and sustained contributions to the ISPlasma conference series.
Since the early years of ISPlasma, Professor Czarnetzki has played a central role in shaping its scientific foundation and long-term development. Through his numerous presentations at ISPlasma, including five plenary, keynote, and invited presentations, as well as six tutorial lectures, Professor Czarnetzki has significantly enriched the education of students and young researchers.
In addition to his scientific contributions, Professor Czarnetzki has also served continuously on the ISPlasma Advisory Committee since 2009, helping to guide the conference's strategic direction and evolution. His achievements were previously recognized with the ISPlasma Prize in 2018.
The ISPlasma Special Recognition Award highlights his sustained excellence in research, education, and service. We congratulate Professor Czarnetzki on this well-deserved recognition.
- Details
Mikhail Sneider at EP5
 During May 4–5, the CRC 1316 had the pleasure of welcoming Prof. Mikhail N. Shneider from Princeton University, USA, as a guest visitor. During his stay, he visited several CRC laboratories and received guided tours at EP2, EP5, and AEPT.
During May 4–5, the CRC 1316 had the pleasure of welcoming Prof. Mikhail N. Shneider from Princeton University, USA, as a guest visitor. During his stay, he visited several CRC laboratories and received guided tours at EP2, EP5, and AEPT.
A highlight of the visit was his seminar talk:
“Combined Laser-Microwave Diagnostics of Gas and Weakly Ionized Plasma”
The seminar attracted researchers and students interested in advanced plasma diagnostics and provided valuable insights into current developments in the field. The visit offered an excellent opportunity for scientific exchange and discussions within the CRC 1316 community.
- Details
Dr. Ibrahim Sadiek Appointed Subcommittee Chair for CLEO 2027
Dr. Ibrahim Sadiek has been appointed Subcommittee Chair for Science & Innovation 13 (S&I 13): Active Optical Sensing within the CLEO conference series, one of the leading international conferences in laser science and photonics, following his service as a subcommittee member in 2026.
S&I 13 covers a wide range of topics in active optical sensing, including spectral, acoustic, and navigational sensing, as well as laser-based diagnostics of plasmas, combustion, and extreme environments. It also includes UV, visible, infrared chemical sensing, and integrated sensing approaches.
As Subcommittee Chair, Dr. Sadiek will lead the selection of committee members, coordinate the identification of invited speakers, and oversee the abstract review process. He will also contribute to shaping the technical program of CLEO 2027.
CLEO 2027 will take place from May 2 - 7, 2027, in Long Beach, California, USA.
Link to CLEO webpage: https://cleoconference.org
- Details
Plasma Science for Teachers
In the 48. Lüscherseminar, organized by TU München, Achim von Keudell gave a lecture on 'Cold Plasmas a key enabling technology of the 21st century' on 18.4.2026 in Zwiesel, Bavaria - Programme.
- Details
Congratulations to Dr. Sascha Chur and Dr. Steijn Vervloedt
 On February 18, 2026, Sascha Chur and Steijn Vervloedt successfully defended their PhD thesis.
On February 18, 2026, Sascha Chur and Steijn Vervloedt successfully defended their PhD thesis.
Steijn worked on project A3 of SFB1316 and focused his thesis on “Plasma-Surface Synergism for Nitrogen Fixation”. Sascha conducted research in project B2 on the synthesis of a copper-based CO2-RR catalyst material. His thesis is entitled “Tuning surface stoichiometry and morphology by atmospheric pressure plasma jet generated reactive oxygen species and short pulsed laser irradiation”.
Steijn was highly praised by his doctoral supervisor Prof. Dr. Achim von Keudell and Sascha by his doctoral supervisor Prof. Dr. Judith Golda.
We wish Sascha and Steijn all the best for the future!
- Details
MGK Kolloquium in Olpe 2026
"When the cats is away the mice will play" is the Motto of the annual MGK kolloquium of the CRC1316. Once a year our ECRs meet at the self-irganized meeting to discuss the successes and hardships of PhD life, without supervision of their PIs. This year, the meeting took place from 25.-27.2. in Olpe.
 The kolloquium began on Wednesday with a classic poster presentation containing recent results of each project. A chance to update each other on the recent progress and practice presentation skills.
The kolloquium began on Wednesday with a classic poster presentation containing recent results of each project. A chance to update each other on the recent progress and practice presentation skills.
On Thursday two workshops were held. The first was on the topic of ElabFTW and data management by Sarah-Johanna Klose and Steffen Schüttler. It included group exercises and exchange on labbook etiquette.
The second workshop on "How to post-doc" was held by Johanna Templ and Maximilian Wohlgemuth from the anorganic Chemistry Chair I of RUB. They gave their insight on the application process for a post doc position and the Dos and Don'ts of the process.
 In the afternoon the ECRs had organized a social event, an outdoor escape room through Olpe. The scavenger hunt-like activity sent the group walking along the Biggesee in best weather to find the fictional Basti who had not been seen at his fictional job in a fictional while. Luckily, in the end he had only gone on a fictional vacation. Good for him!
In the afternoon the ECRs had organized a social event, an outdoor escape room through Olpe. The scavenger hunt-like activity sent the group walking along the Biggesee in best weather to find the fictional Basti who had not been seen at his fictional job in a fictional while. Luckily, in the end he had only gone on a fictional vacation. Good for him!
The day ended with a barbecue.
 On friday, another workshop was held by Jonas Hiepel on the use of AI tools for scientific research including lively exchange amongst the ECRs who had varying levels of experience on the topic.
On friday, another workshop was held by Jonas Hiepel on the use of AI tools for scientific research including lively exchange amongst the ECRs who had varying levels of experience on the topic.
Afterwards, another poster session commenced but this time on current challenges in the lab and beyond. Many discussions were had and problems were solved ( or at least got one step closer to solving).
Overall, the MGK Kolloquium provided the ECRs with an opportunity to discuss their respective PhD projects and opportunities for networking and socialising.
- Details
PI Meeting as Start of CRC 1316’s Third Funding Period
 With the start of the third funding period of CRC 1316, the Principal Investigators (PIs) came together for a two-day strategy meeting. The meeting took place on February 23 and 24 at Ruhr University Bochum.
With the start of the third funding period of CRC 1316, the Principal Investigators (PIs) came together for a two-day strategy meeting. The meeting took place on February 23 and 24 at Ruhr University Bochum.
The discussions focused on reviewing the scientific achievements of the previous funding phase and defining strategic goals for the years ahead. The PIs presented updates on the individual subprojects.
- Details
DFG Extends Funding for Collaborative Research Center SFB 1316
 We are pleased to announce that the German Research Foundation (DFG) has extended funding for the Collaborative Research Center SFB 1316 “Transient Atmospheric Pressure Plasmas – from Plasma to Liquids to Solids” at Ruhr University Bochum for another four years. This decision highlights the scientific importance and innovative potential of our plasma research.
We are pleased to announce that the German Research Foundation (DFG) has extended funding for the Collaborative Research Center SFB 1316 “Transient Atmospheric Pressure Plasmas – from Plasma to Liquids to Solids” at Ruhr University Bochum for another four years. This decision highlights the scientific importance and innovative potential of our plasma research.
Research at the Intersection of Plasma, Catalysis, and Sustainability
In SFB 1316, we study how atmospheric plasmas interact with different materials and how these processes can be precisely controlled. The upcoming funding period will focus on stronger integration of plasma research with catalytic, electrochemical, and biocatalytic processes. This approach opens new pathways for using plasmas in sustainable chemical transformations.
Development of Prototypes for Sustainable Energy Systems
A central goal is to develop initial prototypes for larger-scale systems that efficiently convert renewable energy into chemical energy carriers. This work contributes significantly to the energy transition and the exploration of alternative technologies for CO₂ reduction.
Focus on Modeling and Scalability
We will also expand our efforts in simulation and modeling to better understand the underlying physico-chemical processes. These insights are essential to transition plasma technologies from laboratory research to practical applications.
- Details
International School on Low Temperature Plasma Physics

 In the beginning of October, chosen Master and PhD students within the field of plasma physics, engineering and chemistry had the chance to get in contact with leading experts and lecturers. During the International School on Low Temperature Plasma Physics and the subsequent Master Class on Smart organic coatings by plasmas, fundamentals of plasmas and cutting-edge reserach has been discussed in lectures and during a poster session. Hands-on experience was obtained during three workshops lead by reknown professors from France and Portugal.
In the beginning of October, chosen Master and PhD students within the field of plasma physics, engineering and chemistry had the chance to get in contact with leading experts and lecturers. During the International School on Low Temperature Plasma Physics and the subsequent Master Class on Smart organic coatings by plasmas, fundamentals of plasmas and cutting-edge reserach has been discussed in lectures and during a poster session. Hands-on experience was obtained during three workshops lead by reknown professors from France and Portugal.
Since 28 years, the plasma school is organized by leading experts in the field of plasma physics. This year, the international chair was located at University of Mons in Belgium, while the local organizing team was located at Ruhr University in Bochum. The plasma school has also been funded by the CRC 1316 and the Research Department "Plasmas with Complex Interactions".